|
The fraction remaining is 1/ 2 (0.5 or 50%). If you have a sample of 100 grams of the unstable isotope then after one hour, 50 g would have decayed and 50 g would remain. Say there is an unstable isotope with t 1/2 = 1 hour. Each unstable isotope has a characteristic half-life which is the amount of time that has passed when half of a group of unstable atoms has decayed. Even so, it is possible to describe the behavior of large numbers of atoms using a simple idea. 
The radioactive decay of an atom cannot be hurried up or slowed down in any way. It is impossible to predict when a single unstable atom will decay. Individual unstable atoms decay completely randomly. One way to write this law is in terms of something called the half-life of the decay. Measure this decrease in radiation and discovered a simple law ofĮxponential decay to describe the decrease. 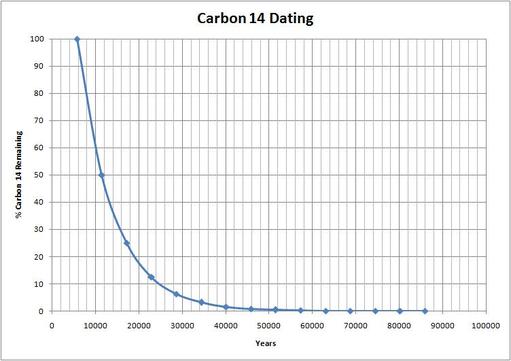
Investigations into atomic structure) found that his sources of Radioactive Half-life See also: PhET Radioactive Dating GameĪround the year 1900 Ernest Rutherford (a key figure in early
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
